Want to test what you know?
Try our free Grade 8 Chemistry quiz — 25 questions, instant results, no sign-up.
Periodic Table Made Easy: Groups, Periods & How to Remember Elements
The periodic table can seem intimidating—118 elements, strange symbols, confusing trends. But it's actually a beautifully organized system that reveals the hidden patterns of chemistry.
In this guide, we'll demystify the periodic table, help you remember elements with clever mnemonics, and show you the patterns that make chemistry predictable.
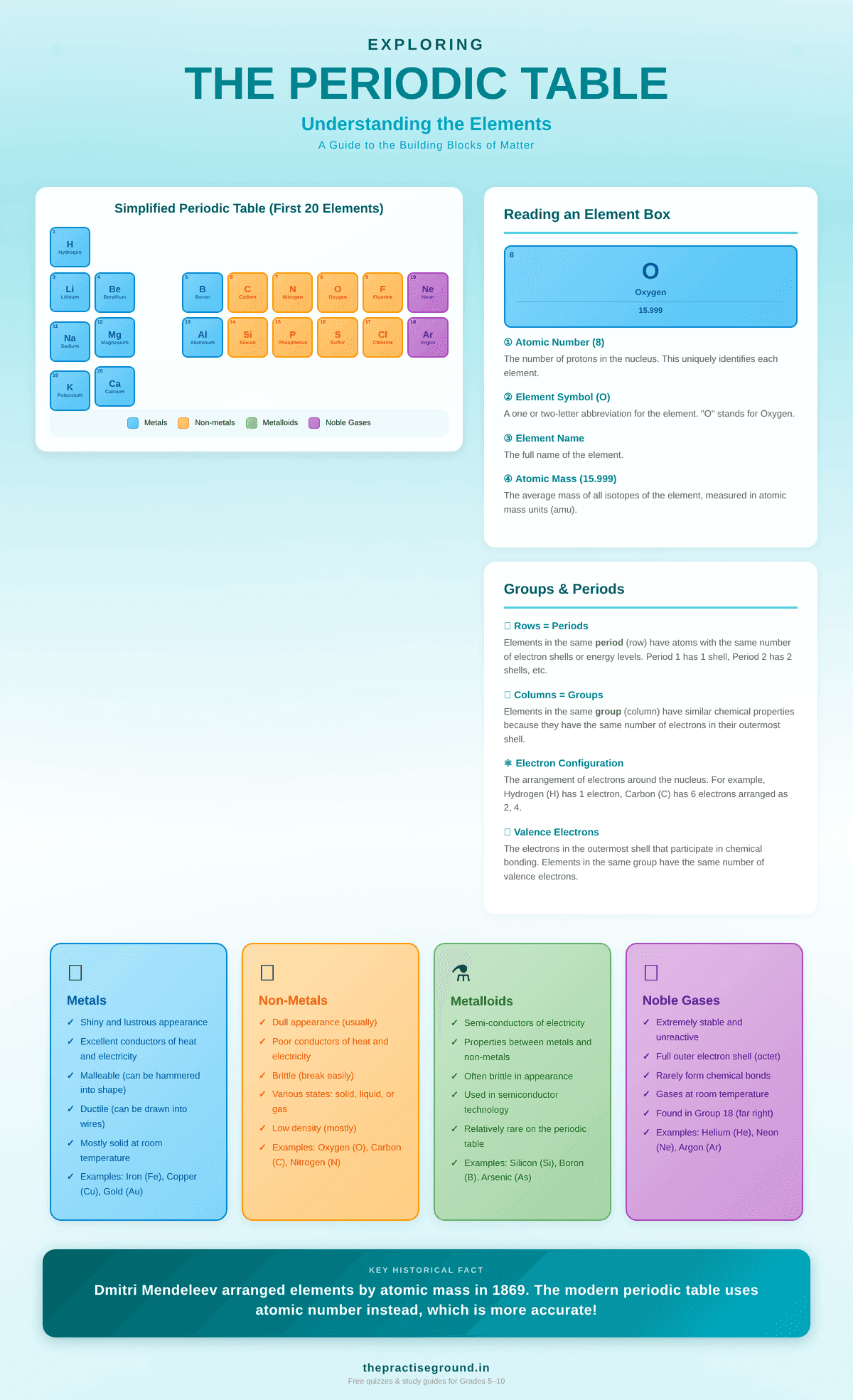
What Is the Periodic Table?
The periodic table is a systematic arrangement of all known chemical elements organized by their atomic number and properties. The word "periodic" means the properties repeat in a regular pattern—hence the table's name.
Why is it important?- It shows relationships between elements
- Predicts element properties
- Helps you understand chemical reactions
- Essential for CBSE, ICSE, and IB chemistry courses
How Is the Periodic Table Organized?
By Atomic Number (Most Important)
Elements are arranged from left to right, top to bottom, by increasing atomic number (number of protons in the nucleus).
By Periods (Horizontal Rows)
The period number indicates how many electron shells (or energy levels) an atom has.
By Groups (Vertical Columns)
The group number indicates how many valence electrons (electrons in the outermost shell) an atom has.
This is why vertical columns have similar properties—they have the same number of valence electrons!
Important Groups
Group 1: Alkali MetalsCommon Elements You Must Remember
The First 20 Elements (Absolutely Critical)
Memorize these in order:
CODEBLOCK0 Mnemonic for first 20:"Happy Henry Likes Beer But Could Not Obtain Four Nests; Nancy Mg'd Always Sigh Piteously Swearing Cloyingly, Arguing Kindly Canned"
(Break it down into 10-element chunks if this is too long!)
Important Elements Beyond First 20
Periodic Trends: The Patterns That Make Chemistry Predictable
Understanding trends is more valuable than memorizing individual elements. Trends help you predict properties!
1. Atomic Radius (Size of the Atom)
Trend 1: Decreases across a period (left to right)Why? As you move right, you add more protons, which pull electrons closer.
Trend 2: Increases down a groupWhy? Each element has an additional electron shell, making atoms larger.
Visual: The biggest atoms are in the bottom-left (Francium), the smallest are in the top-right (Fluorine).2. Ionization Energy
Definition: Energy required to remove an electron from an atom. Trend 1: Increases across a period (left to right)Why? Atoms with more protons hold electrons more tightly.
Trend 2: Decreases down a groupWhy? Valence electrons are farther from the nucleus, easier to remove.
Real-world: Group 1 metals have low ionization energy (easy to remove 1 electron, forming +1 ions).3. Electronegativity
Definition: An atom's tendency to attract electrons in a chemical bond. Trend 1: Increases across a period (left to right) Trend 2: Decreases down a group Extremes: Fluorine is the most electronegative element. Francium is the least electronegative metal.4. Metallic Character
Trend 1: Decreases across a period (left to right)Metals → Semimetals → Nonmetals
Trend 2: Increases down a group Staircase line: The periodic table has a diagonal staircase line (B, Si, Ge, As, Sb, Te, At) separating metals from nonmetals.Quick Recap: Periodic Table Organization
| Concept | Left to Right (Across Period) | Top to Bottom (Down Group) |
|---|---|---|
| Atomic Radius | Decreases | Increases |
| Ionization Energy | Increases | Decreases |
| Electronegativity | Increases | Decreases |
| Metallic Character | Decreases | Increases |
Memory Tricks for Chemistry Students
Trick 1: Using Element Symbols as Stories
Instead of just memorizing "Na, Mg, Al, Si...", create a story:
"Na (an old lady) took her Mg (magnesium salt bath), while Al (aluminum foil) covered her Si (silicon valley estate)..."
Silly stories are memorable!
Trick 2: Group Mnemonics
Group 1 (Alkali Metals): LiNaKRbCsFr = "LittlNa girls Keep Raising Cstunning Friends" Group 17 (Halogens): FCBrI (At is too radioactive to worry about) = "Flunky Clowns Bring Ice"Trick 3: Color-Code Your Periodic Table
Visual memory is stronger than text memory!
Mendeleev's Periodic Table vs. Modern Periodic Table
Mendeleev (1869)
Dmitri Mendeleev organized elements by increasing atomic weight and left gaps for undiscovered elements (which turned out to exist!). Brilliant prediction.
Limitation: Didn't explain why elements followed this pattern.Modern Periodic Table
Organized by atomic number (not weight), which perfectly explains the periodic pattern using electron configurations.
Advantage: Explains the "why" behind chemistry.Real-Life Applications: Where Elements Are Used
Indian Context (Agriculture, Industry, Medicine)
Try This: Element Challenges
- Predict the property: "Which element would you expect to be more reactive: Chlorine (Group 17, Period 3) or Bromine (Group 17, Period 4)?"
Exam Questions: CBSE/ICSE Pattern
Q1: In which period and group is oxygen located?A: Period 2 (2 electron shells), Group 16 (6 valence electrons).
Q2: Why is fluorine more electronegative than chlorine?A: Fluorine is smaller and higher in the group. Its valence electrons are closer to the nucleus and held more tightly.
Q3: Arrange in order of increasing atomic radius: Cl, S, PA: Cl < S < P (moving left in the same period, atomic radius increases).
Q4: Why are noble gases unreactive?A: They have complete valence shells (8 electrons, except He), so they don't need to gain, lose, or share electrons.
Q5: What is the relationship between group number and valence electrons in the main-group elements?A: The group number indicates the number of valence electrons (e.g., Group 13 has 3 valence electrons, Group 17 has 7).
FAQ: Periodic Table
Q: Why are there gaps and strange arrangements in the transition metals?A: Transition metals have complex electron configurations. For d-block elements, electrons are filling inner shells (d orbitals), which creates unusual patterns. This is beyond basic chemistry but important in advanced courses.
Q: Why is hydrogen placed separately from Group 1?A: Although hydrogen has 1 valence electron (like Group 1), it's fundamentally different. It can form covalent bonds and doesn't behave like alkali metals. Some modern periodic tables place it separately.
Q: How many elements have been discovered so far?A: 118 confirmed elements. Elements beyond Uranium (92) are synthetic, created in laboratories, and highly unstable with extremely short half-lives.
Q: What makes noble gases "noble"?A: The term "noble" reflects their unreactive nature—they don't "lower themselves" by reacting with other elements (mostly!). It's a historical term that stuck.
Q: Can I predict chemical formulas using the periodic table?A: Yes! For main-group elements, the group number often predicts charge: Group 1 forms +1, Group 2 forms +2, Group 17 forms -1, Group 16 forms -2, etc.
Next Steps
Now that you understand the periodic table, explore related topics:
The periodic table is your chemistry handbook. Return to it whenever you need to predict element behavior—that's exactly what Mendeleev intended. Master it, and you'll find chemistry becomes far more logical and predictable.
Taking a quick break? Test yourself!
Pause and try a quick Grade 8 Chemistry quiz — see how much you've picked up so far.