Want to test what you know?
Try our free Grade 7 Chemistry quiz — 25 questions, instant results, no sign-up.
Acids, Bases and Salts — Properties, pH Scale & Everyday Examples
Acids and bases are everywhere in your daily life—from the lemon juice in your daal to the soap you use for washing clothes. Understanding their properties helps you understand chemistry, cooking, cleaning, and even your body's health.
In this guide, we'll explore acids, bases, and salts with properties, the pH scale, and real-world examples that matter for CBSE, ICSE, and IB examinations.
What Are Acids?
An acid is a substance that:- Tastes sour
- Turns blue litmus paper red
- Releases hydrogen ions (H⁺) in solution
- Reacts with metals to produce hydrogen gas
- Reacts with bases to form salts and water
Properties of Acids
- Sour taste (but never taste lab chemicals!)
- Turn blue litmus red
- Conduct electricity (because they contain ions)
- Corrosive (can damage materials and skin)
- React with metals: Acid + Metal → Salt + Hydrogen gas
Common Acids and Examples
| Acid | Formula | Source | Use |
|---|---|---|---|
| Hydrochloric Acid | HCl | Stomach | Digestion |
| Sulfuric Acid | H₂SO₄ | Car batteries | Battery acid |
| Acetic Acid | CH₃COOH | Vinegar | Cooking, pickling |
| Citric Acid | C₆H₈O₇ | Lemons, lime | Preservative |
| Lactic Acid | C₃H₆O₃ | Yogurt | Fermented foods |
| Nitric Acid | HNO₃ | Fertilizers | Agriculture |
Acids in Indian Context
What Are Bases?
A base is a substance that:Properties of Bases
Common Bases and Examples
| Base | Formula | Source | Use |
|---|---|---|---|
| Sodium Hydroxide | NaOH | Drain cleaner | Industrial |
| Ammonia | NH₃ | Fertilizer, cleaner | Farming, cleaning |
| Calcium Hydroxide | Ca(OH)₂ | Slaked lime | Whitewash, construction |
| Potassium Hydroxide | KOH | Soap | Cleaning |
| Baking soda | NaHCO₃ | Kitchen | Cooking, cleaning |
Bases in Indian Context
Alkalis: A Special Type of Base
An alkali is a base that's soluble in water. Not all bases are alkalis—for example, calcium hydroxide is a base, but it's slightly soluble (not a true alkali).
Common alkalis: NaOH, KOH, NH₃ (all dissolve in water)
What Are Salts?
A salt is the product formed when an acid reacts with a base. General reaction: Acid + Base → Salt + WaterExamples of Salts
| Salt | Formula | Source | Use |
|---|---|---|---|
| Sodium Chloride | NaCl | Sea salt | Cooking, de-icing |
| Calcium Carbonate | CaCO₃ | Limestone | Construction |
| Potassium Nitrate | KNO₃ | Fertilizer | Agriculture |
| Copper Sulfate | CuSO₄ | Fungicide | Farming |
| Magnesium Sulfate | MgSO₄ | Epsom salt | Medicine, agriculture |
| Sodium Bicarbonate | NaHCO₃ | Baking soda | Cooking |
Properties of Salts
Salts in Indian Daily Life
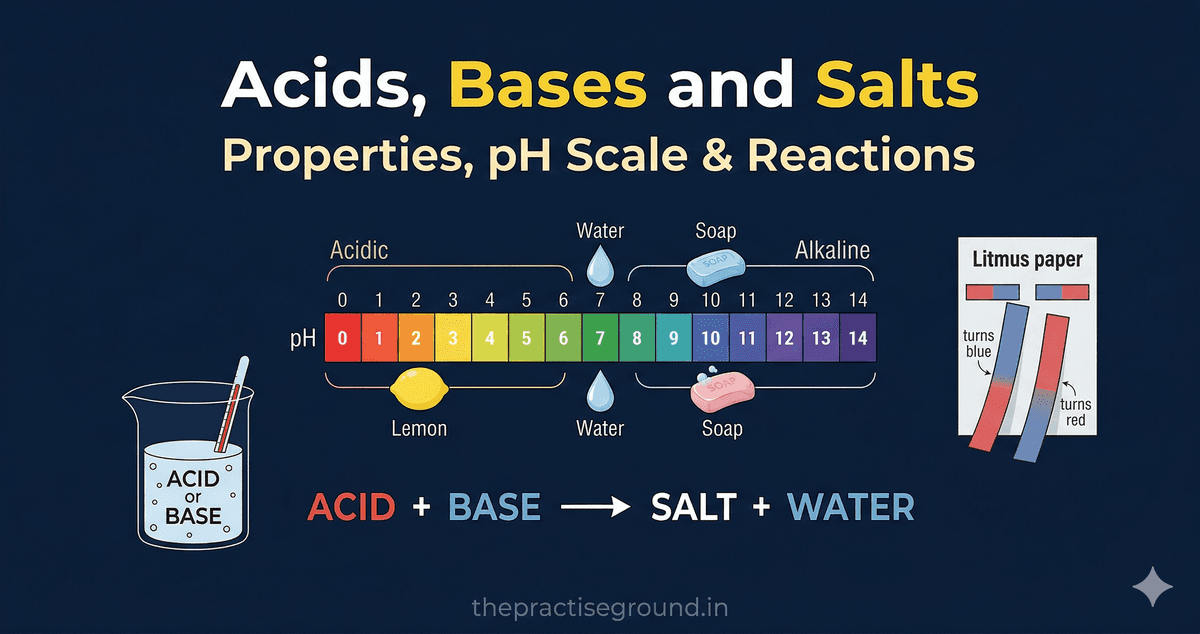
The pH Scale: Measuring Acidity and Basicity
The pH scale measures how acidic or basic a solution is.
CODEBLOCK0Understanding pH Values
Everyday pH Examples
| Substance | pH | Category |
|---|---|---|
| Stomach acid | 1.5-3.5 | Strongly acidic |
| Lemon juice | 2-3 | Acidic |
| Vinegar | 2-3 | Acidic |
| Milk | 6.5-7 | Slightly acidic to neutral |
| Pure water | 7 | Neutral |
| Human blood | 7.35-7.45 | Slightly basic |
| Baking soda solution | 8-9 | Slightly basic |
| Ammonia solution | 11-13 | Basic |
| Drain cleaner | 13-14 | Strongly basic |
pH in Human Body
Your body maintains very specific pH values:
Disruption in these pH levels can cause health problems.
Indicators: How to Detect Acids and Bases
Indicators are substances that change color based on pH.Common Indicators
| Indicator | Acidic | Neutral | Basic |
|---|---|---|---|
| Litmus Paper | Red | Purple | Blue |
| Methyl Orange | Red | Orange | Yellow |
| Phenolphthalein | Colorless | Colorless | Pink |
| Turmeric | Yellow | Yellow | Brown/Black |
| Red Cabbage | Purple/Pink | Purple | Yellow/Green |
Natural Indicators from Indian Kitchen
These work surprisingly well for simple pH testing!
Neutralization Reaction
When an acid and base react, they neutralize each other, producing a salt and water.
Equation: Acid + Base → Salt + Water Example: CODEBLOCK1Real-Life Neutralization Examples
Types of Salts Based on pH
Not all salts are neutral! Some are acidic or basic:
Neutral Salts
Acidic Salts
Why acidic? The cation (NH₄⁺ or Fe³⁺) hydrolyzes, releasing H⁺ ions.
Basic Salts
Why basic? The anion (CO₃²⁻ or CH₃COO⁻) hydrolyzes, releasing OH⁻ ions.
Quick Recap: Acids vs Bases vs Salts
| Property | Acids | Bases | Salts |
|---|---|---|---|
| Taste | Sour | Bitter | Various |
| Litmus | Red | Blue | No change |
| pH | < 7 | > 7 | Can vary |
| H⁺ ions | Release | Absorb | None (usually) |
| Reaction | With bases → salt + water | With acids → salt + water | Can react with acids/bases |
Try This: Practical Experiments
Exam Questions: CBSE/ICSE Pattern
Q1: What is the pH of pure water?A: pH = 7 (neutral). Equal concentration of H⁺ and OH⁻ ions.
Q2: Write the reaction when an acid reacts with a base.A: Acid + Base → Salt + Water Example: HCl + NaOH → NaCl + H₂O
Q3: How would you distinguish between an acid and a base using litmus paper?A: Acids turn blue litmus red. Bases turn red litmus blue.
Q4: Name three natural indicators used to test pH.A: Red cabbage, turmeric, hibiscus, henna (any three).
Q5: Why is pH control important in agriculture?A: Different crops grow best at specific pH values. Acidic soil requires lime (calcium hydroxide) to neutralize. Basic soil requires sulfur additions. Maintaining proper pH ensures nutrient availability and crop growth.
FAQ: Acids, Bases and Salts
Q: Is water acidic or basic?A: Pure water is neutral (pH 7). However, rainwater is slightly acidic (pH ~5.6) because it dissolves CO₂ from the atmosphere, forming weak carbonic acid.
Q: Can a salt be acidic?A: Yes! Salts formed from weak bases and strong acids are acidic. For example, ammonium chloride (NH₄Cl) is acidic because NH₄⁺ hydrolyzes to release H⁺.
Q: Why do antacids work?A: They contain bases (like Mg(OH)₂ or Al(OH)₃) that neutralize excess stomach acid (HCl). The neutralization reaction reduces acidity.
Q: What happens if you mix a strong acid with a strong base?A: They neutralize each other completely, producing a neutral salt and water. If quantities are equal, the resulting solution has pH 7.
Q: Are all bases called alkalis?A: No. Alkalis are bases that dissolve in water. For example, NaOH is an alkali (soluble base), but Ca(OH)₂ is a base but not a strong alkali (less soluble).
Next Steps
Now that you understand acids, bases, and salts, explore related topics:
Acids and bases are fundamental to chemistry, biology, and everyday life. Understanding them opens doors to understanding medicine, cooking, agriculture, and environmental science. Master this topic, and you'll have a strong foundation for all advanced chemistry.
Taking a quick break? Test yourself!
Pause and try a quick Grade 7 Chemistry quiz — see how much you've picked up so far.