Want to test what you know?
Try our free Grade 8 Chemistry quiz — 25 questions, instant results, no sign-up.
Chemical Reactions & Equations: Types, Balancing & Examples
Chemical reactions happen everywhere—from rusting iron to digestion in your stomach. Understanding how to represent and balance these reactions is fundamental to chemistry and crucial for CBSE Chapter 1, ICSE, and IB exams.
In this guide, we'll learn how to write balanced equations, understand different reaction types, and master the concepts that appear repeatedly on exams.
What Is a Chemical Reaction?
A chemical reaction is a process where substances (reactants) transform into different substances (products) with different properties.
Key features:- Bonds break in reactants
- Bonds form in products
- Atoms rearrange but are never created or destroyed
- Energy is released or absorbed
Chemical Equations: Representing Reactions
A chemical equation uses chemical formulas and symbols to represent a reaction.
Parts of a Chemical Equation
CODEBLOCK0 Breaking it down:Physical States
Letters in parentheses show physical states:
CODEBLOCK1Balancing Chemical Equations: The Golden Rule
Law of Conservation of Mass: Matter is not created or destroyed in a chemical reaction. Atoms on the left (reactants) must equal atoms on the right (products).The Balancing Method (Hit and Trial)
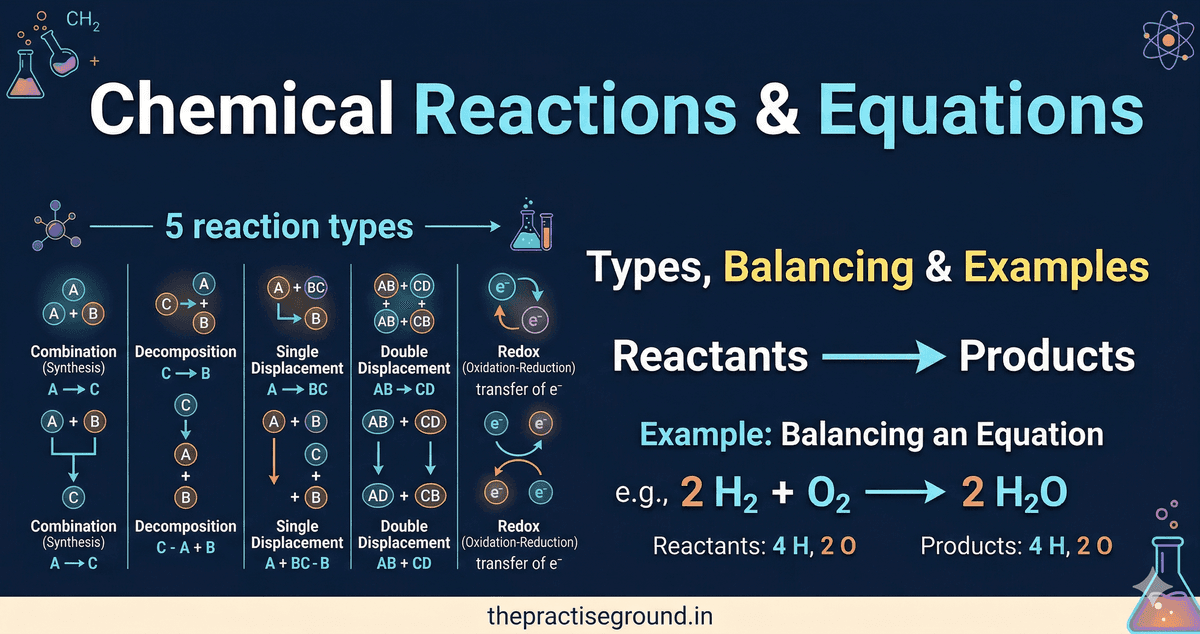
Step 1: Count atoms of each element on both sides Step 2: Identify which elements are unbalanced Step 3: Add coefficients to balance (never change subscripts!) Step 4: Recount to verify all atoms balanceExample: Balancing H₂ + O₂ → H₂O
Step 1: Count atomsUnbalanced: 2H₂ + O₂ → H₂O
Both H and O are unbalanced.
Step 3: Add coefficientsTry: 2H₂ + O₂ → 2H₂O
All atoms balance! The balanced equation is: CODEBLOCK2
This represents: Two hydrogen molecules react with one oxygen molecule to produce two water molecules.
More Practice Examples
Example 1: Burning of CarbonUnbalanced: C + O₂ → CO₂
Unbalanced: Fe + O₂ → Fe₂O₃
Balance iron first:
Types of Chemical Reactions
Different reactions follow patterns. Recognizing these patterns helps you predict products.
1. Combination Reactions (Synthesis)
Pattern: A + B → ABTwo or more reactants combine to form one product.
Examples: CODEBLOCK3 Real-world: Burning fuels, rusting metals, plant respiration2. Decomposition Reactions
Pattern: AB → A + BOne compound breaks into two or more simpler substances.
Examples: CODEBLOCK4 Real-world: Decomposition reactions are used to extract metals from ores.3. Displacement Reactions (Single Displacement)
Pattern: A + BC → AC + BOne element displaces another element from a compound.
Examples: CODEBLOCK5 Reactivity series: Some elements are more reactive and can displace less reactive ones. Common reactivity series (most to least reactive): K > Na > Ca > Mg > Al > Zn > Fe > Cu > Ag Rule: An element can only displace a less reactive element. Real-world: Used in extraction of metals.4. Double Displacement Reactions
Pattern: AB + CD → AC + BDIons exchange between two compounds.
Examples: CODEBLOCK6 Real-world: Precipitation reactions, acid-base neutralization.5. Redox Reactions (Oxidation-Reduction)
Definition: Reactions where electrons are transferred between atoms. Oxidation: Loss of electrons (or gain of oxygen) Reduction: Gain of electrons (or loss of oxygen) Examples: CODEBLOCK7 Memory aid: OIL RIG6. Combustion Reactions
Pattern: Fuel + O₂ → CO₂ + H₂O + EnergyFuel burns in oxygen, releasing energy.
Examples: CODEBLOCK8 Real-world: Burning natural gas, petrol, candles, cooking.Quick Recap: Reaction Types
| Type | Pattern | Example | Result |
|---|---|---|---|
| Combination | A+B→AB | 2Na+Cl₂→2NaCl | Single product |
| Decomposition | AB→A+B | 2H₂O→2H₂+O₂ | Multiple products |
| Displacement | A+BC→AC+B | Zn+CuSO₄→ZnSO₄+Cu | Element swap |
| Double Displacement | AB+CD→AC+BD | AgNO₃+NaCl→AgCl+NaNO₃ | Ion exchange |
| Redox | Electron transfer | 2Cu+O₂→2CuO | Oxidation-Reduction |
| Combustion | Fuel+O₂→CO₂+H₂O | CH₄+2O₂→CO₂+2H₂O | Burning |
Representation of Chemical Reactions
Skeleton Equation
Shows what reacts and what forms (unbalanced): CODEBLOCK9
Balanced Equation
Shows correct number of molecules (balanced): CODEBLOCK10
Complete Ionic Equation
Shows all ions in solution: CODEBLOCK11
Energy in Chemical Reactions
Exothermic Reactions
Release energy (usually heat and light).
Examples: Combustion, neutralization, rusting Equation: Reactants → Products + EnergyEndothermic Reactions
Absorb energy (usually heat).
Examples: Photosynthesis, decomposition, melting ice Equation: Reactants + Energy → ProductsReal-Life Applications: Indian Context
Common Exam Mistakes to Avoid
- Changing subscripts: Only coefficients change, never subscripts
- Unbalanced equations: Always verify both sides
- Forgetting states: Include (s), (l), (g), (aq)
- Wrong products: Know the products before balancing
- Over-complicating: Use smallest whole number coefficients
Try This: Balancing Practice
Exam Questions: CBSE/ICSE Pattern
Q1: What is a balanced chemical equation? Why is balancing necessary?A: A balanced equation has equal numbers of each atom on both sides. Balancing is necessary because of the Law of Conservation of Mass—atoms are neither created nor destroyed.
Q2: Balance the equation: Fe + Cl₂ → FeCl₃A: 2Fe + 3Cl₂ → 2FeCl₃ (Count: Left has 2Fe, 6Cl; Right has 2Fe, 6Cl ✓)
Q3: Classify the reaction: 2PbO → 2Pb + O₂A: Decomposition reaction (one compound breaks into simpler substances).
Q4: Using the reactivity series, predict the reaction: Zn + FeSO₄ → ?A: Zn is more reactive than Fe, so Zn displaces Fe: Zn + FeSO₄ → ZnSO₄ + Fe
Q5: What is oxidation and reduction? Give examples.A: Oxidation is loss of electrons; Reduction is gain of electrons. Example: 2Cu + O₂ → 2CuO (Cu is oxidized, O is reduced)
FAQ: Chemical Reactions and Equations
Q: What's the difference between a physical and chemical change?A: Physical change doesn't form new substances (melting, dissolving, color). Chemical change forms new substances with different properties (burning, rusting, digestion).
Q: Why do we balance equations?A: To represent the actual molecular ratio in which reactants combine. Also, balanced equations are needed for stoichiometry calculations.
Q: Can a reaction be both combination and redox?A: Yes! Most combination reactions involving nonmetals with oxygen are both combination and redox (electrons are transferred).
Q: What's the difference between decomposition and combustion?A: Combustion specifically involves burning in oxygen. Decomposition can happen in absence of oxygen (example: 2PbO → 2Pb + O₂ under heat).
Q: How do I know if a precipitate will form?A: Use solubility rules. Most carbonates, hydroxides, and halides of heavy metals form precipitates. You'll have a table for this in your textbook.
Next Steps
Now that you understand chemical reactions, explore related topics:
Chemical equations are the language of chemistry. Master them, and you've unlocked the ability to communicate chemical ideas precisely. This skill is essential for success in chemistry at any level. Good luck with your exams!
Taking a quick break? Test yourself!
Pause and try a quick Grade 8 Chemistry quiz — see how much you've picked up so far.